Origin Pharma Packaging is dedicated to excellence, which benefits not only our pharmaceutical and healthcare clients with quality pharmaceutical packaging, wellbeing product packaging, and medical cannabis packaging but also the global consumer market as a whole.
One specialist addition to our ongoing service enhancement is a multi-million-pound investment programme at our Pharmaceutical Packaging, Logistics & Innovation Centre to offer premier cleanroom manufacturing services.
This major development has involved the design and installation of a state-of-the-art cleanroom production facility that strictly adheres to ISO 7 class 10,000 and ISO 8 class 100,000, which each ensure the reliable prevention and control of airborne particles. Achieving these standards is no mean feat, as the cleanroom environment must comply with the following specifications at all times with no exceptions:


Ideal for Wide-Ranging Requirements
Some business owners, such as those who are relatively new to the healthcare sector or have recently launched a start-up, may think that this kind of cleanroom manufacturing facility is only for large-scale projects. However, thanks to flexibility being integrated at its very core, our cleanroom can also supply small product runs for pharmaceutical and healthcare companies based around the world and deliver them on a global scale.

Dedicated Cleanroom Environment
In accordance with ISO 14644-1, an ISO 1 environment is the cleanest level possible and ISO 9 is at the other end of the scale, although it will still be significantly cleaner than a normal production room. The most common classes of cleanroom are ISO 7 and ISO 8, which must also meet industry-specific and regional standards such as EU GMP guidelines A, B, C and D, with pharmaceutical products typically being Class C (requiring an ISO 7 cleanroom) and Class D (ISO 8).

Distinct Standards. Leading Technologies.
The creation of a cleanroom for pharmaceutical manufacturing is a highly meticulous undertaking. Whilst its name is self-explanatory, there are numerous criteria that need to be incorporated into the environment in order for it to qualify as a cleanroom.
For instance, the design and testing stages revolve around the principles of ISO 14644, consistently complying to distinct standards in terms of the quantity and size of particles per volume of air.

Origin’s Jar, bottle and cap manufacturing cleanroom facility in East Yorkshire achieves ISO 7 whilst inactive and ISO 8 when in operation, which results in the following conditions:
- A maximum concentration of 352,000 particles per cubic metre of >= 0.5micron (10,000/cubic ft) at rest and
- A maximum of 3,520,000 particles per cubic metre of >=0.5 micron (100,000/cubic ft) when operational.
Unparalleled Air Control
In order to maintain a compliant cleanroom for pharmaceutical manufacturing, the air pressure within it must be higher than its ambient surroundings. The result is a unidirectional outgoing airflow, with an incoming airflow that’s strictly monitored by our advanced systems as precise cleanroom packaging manufacturers.

The air that does enter the cleanroom is HEPA filtered, removing more than 99% of particles larger than 0.3 µm. Aside from controlling the particles, our cleanroom engineers also perform temperature and humidity control to keep the production process stable and reliable.
A Solution-Driven Approach
Our primary packaging solution specialists appreciate that cleanroom manufacturing processes are highly complex, which is why we work alongside your team to determine the most effective method of production. This could be the use of blow moulding, injection moulding, or another suitable option, as we believe that a one-size-fits-all mentality should never be applied to the manufacture of pharmaceutical and healthcare primary packaging solutions or cleanroom manufacturing services.


Primary Packaging
If you require PET bottles, PP bottles, HDPE bottles, or LDPE bottles, you can also rest assured that our facility has a GMP Class C cleanroom of no less than 1,200 square metres. This specifically air-pressured environment can implement additional treatments such as gamma radiation for medically sterile primary packaging products, which is perfect for clients who require an aseptic automated filling facility.
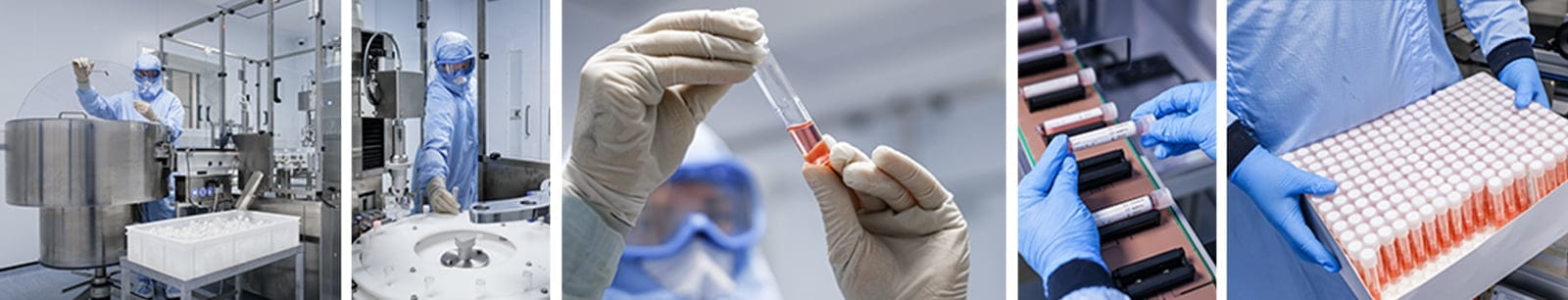
Choose Origin
With over 60 years of experience working with many of the world’s leading brand owners, we’re proud to offer our customers a range of custom-made plastic packaging and glass packaging solutions developed in our state-of-the-art cleanroom in the U.K.
Our facilities enable us to manufacture innovative thermoformed solutions, such as barrier packaging, engineered to maximise revenue, reduce cost, and increase your speed to market. No matter what you need when it comes to a cleanroom manufacturing facility for production, Origin Pharma Packaging will deliver truly exceptional cleanroom manufacturing services.




