Packaging for Medicinal Cannabis
We offer customers a remarkable range of versatile medicinal packaging solutions that respond to the unique needs of the global medicinal cannabis marketplace. We facilitate our customer’s vision to succeed by providing a wide scope; from concept design through to market solutions, supported by extensive global resources.
The Origin Jar, the world’s favourite dried flower jar, is produced in our cleanroom manufacturing facility in the UK. From this starting point, we have broadened our range to cover most primary packaging needs of the medicinal cannabis market.
Origin specialises in medicinal cannabis packaging, ranging from a large amount of stock products which are ready for call-off, to custom packaging, designed and created by our in-house packaging experts. Our team have the knowledge to advise on the correct choice of materials for your packaging to achieve the best results for your target customers.

The cleanroom consists of the following specification:
- Classification: ISO7 class 10, 000 to ISO8 class 100, 000
- Compliant to ISO14644 | ISO9001
- ISO 15378 | BRC
- GMP accredited
- All manufacturing processes are electrically driven
We utilise a range of manufacturing techniques from stretch blow moulding to injection moulding. We will work alongside you to determine the best option for the desired packaging outcome you are looking to achieve.
UK Medicinal Cannabis Packaging Manufacturing Facilities
The Origin Jar is made in our cleanroom manufacturing facilities (GMP accredited), enabling us to create industry-leading packaging, compliant with strict pharmaceutical standards. Origin’s market-leading production environment has a range of production techniques, making it one of the most flexible cleanroom packaging manufacturing facilities in the UK.
Our vast amount of experience in pharmaceutical packaging and devices aligns with the development of new and existing medicinal cannabis products.
Custom Packaging Design

Medicinal Cannabis Packaging
Dried cannabis flower packaging
- Pharmaceutical grade HDPE jars with compatible child resistant closures
- Certified to ISO8317 and 16CFR
- Produced in any custom colour to order
Dried cannabis flower packaging
- Amber glass jar with compatible child resistant closures
- Wide neck for easy dispensing
- Holds up to 10g of dried cannabis flower
Tincture and liquid dose packaging
- Tamper Evident and Child Resistant (or ‘child proof’) pipette closure
- Choose glass or plastic dropper bottles in different sizes and colours for total delivery system
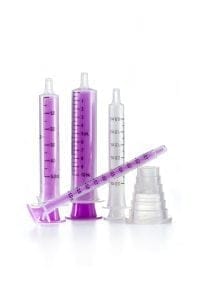
Tincture and liquid dose packaging
- Available in four sizes, all with clear graduation marks for easier measurement
- Child resistant closures available integrated or separate
- Confirms to BS3221:7 and CE marked
Tincture and liquid dose packaging
- Compact size with a cosmetic-style aesthetic
- Range of sizes and neck finishes
- Certified to 16CFR
- Available in a range of colours
Distillate packaging
- Amber Type III glass to protect contents from UV light
- Wide neck for easy access
- Match with an aluminium closure and pressure sensitive liner for a complete solution
Medical cannabis (medicinal) is fast becoming a form a medication that is proving to be very powerful which can have positive effects on the human body.
Cannabis is a plant and comes from the cannabaceae family and is made up of a number of chemicals, which are biologically based. The common compounds that are prominent within are delta-9-tetrahydrocannabinol (THC) and cannabidiol (CBD).
The only cannabis-based product allowed to market and has been approved by the FDA is the drug, Epidiolex, a drug which has been developed to assist people with severe epilepsy. It has been passed as the CBD (not THC) based product is effective for its intended use of supressing seizures.
It is important to understand that THC is a constitute of cannabis and causes the “high”, therefore THC is a potential cause for unknown outcomes for the patient.
Cannabidoid
Is one of several chemicals found in cannabis, also known as the marijuana plant. Cannabidoids can range from THC and CBD which both have different-potential effects on the body.
THC
Is the major psychoactive component of hemp. THC can be extracted and is used in the FDA approved drug Dronabinol, which is to help treat nausea associated with cancer medicines. Studies have also found that THC can potentially help improve memory.
CBD
Cannabidiol is up to 40% of hemp’s extract. CBD can be consumed orally via liquid or can be vaped. CBD does not have the same psychoactive effects as THC. A CBD drug has been given FDA approval for epilepsy disorders.
Cannabis based productions for medicinal use
There are a number of cannabis based products being developed for medicinal purposes. One major drug development which has had FDA approval is Epidiolex. The FDA is continually analysing the regulatory frameworks for products that contain cannabis.
Synthetic cannabidoids for medicinal use
Synthetic developments with cannabidiods have a stronger effect on the body and can be damaging, in comparison to its natural counterparts. However, if developed correctly and used appropriately, synthetic cannabidiods could have a therapeutic and desired effect.
Non-medicinal CBD products
Non-medicinal CBD oil comes from industrial hemp. These products have become popular as they don’t create a ‘high’, and may help in reducing inflammation or pain.
Non-medicinal cannabis
Cannabis, also known as marijuana, is used for recreational purposes and has both creative psychoactive and physiological effects. It is illegal in most countries although recent years have see some countries and USA federal states legalise it.
Non-medicinal synthetic cannabidoids
Are specifically designed and are not naturally derived from the hemp plant. These ‘designer’ drugs- have been created to avoid legal restrictions within specific countries.
Origin is involved with a number of major medical cannabis-based products and is strategically placed across Europe, North America and Australasia to support new projects which are in early stage development. With our packaging knowledge and supply chain model we enable your product to go from development to commercial in record speeds.