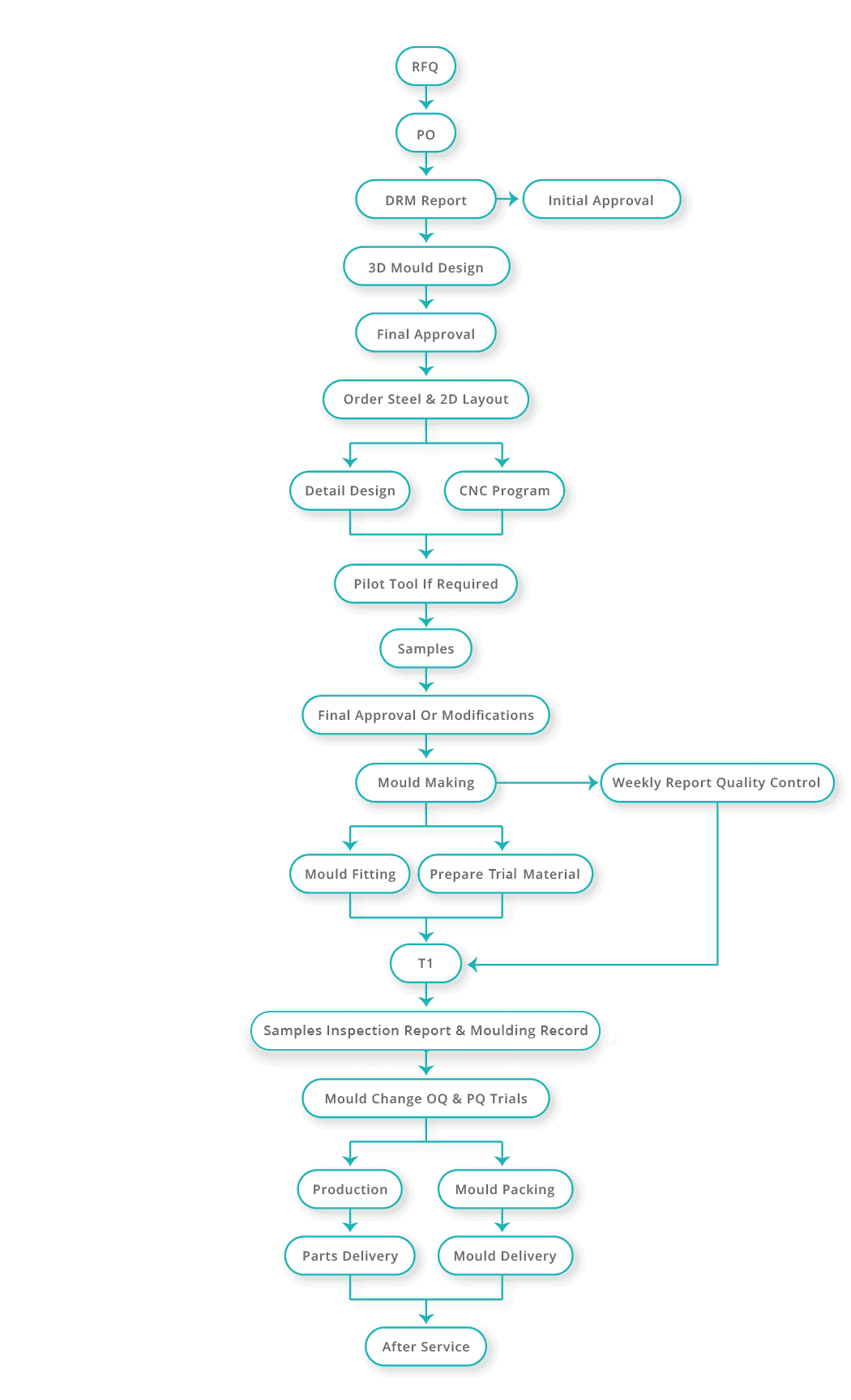
Our comprehensive service and delivery of quality, primary pharmaceutical packaging continues with our agility in tooling design and development.
We are committed to a program of continuous refinement and investment in tool design and material research. Origin embrace all the core moulding processes in our cleanroom manufacturing environment, catering to development/pilot tooling right through to medium cavitation tooling. The facility hosts a number of automatic assembly machines and includes a class 5 aseptic filling facility, enabling us to provide a closed loop mould-and-fill service in one location.
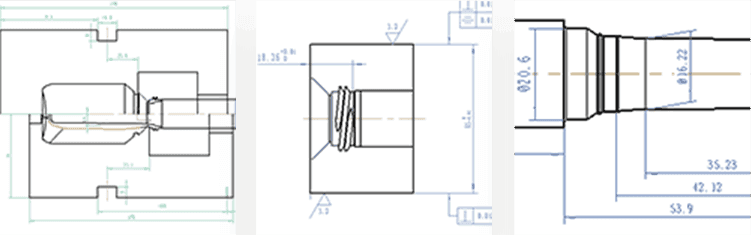
Origin have earned a reputation in mould optimisation. This activity goes beyond the initial tooling development phase, focussing on the intricacies of tooling design, how to optimise the cycle time, the control parameters within the mould, and hybrid features offering flexibility often overlooked in conventional design processes.
Research and Development Department
Origin have brought together a team within the Research and Development department who drive precision and accuracy to develop and manufacture moulds that provide years of reliable operation. Our tooling design and development team have cross-industry experience and a reputation for bringing techniques from parallel markets into our pharmaceutical and healthcare focus.

We support your development from initial Solidworks design, modelling, 3D printing, pilot development – through to validation and commercial production.
Production Techniques and Capabilities
Our class 7 cleanroom moulding and assembly facility is the ideal space for your pharmaceutical packaging project which is supported by a range of plastic packaging manufacturing techniques:
- Injection moulding
- Injection blow moulding
- Injection stretch blow moulding
- Automatic assembly

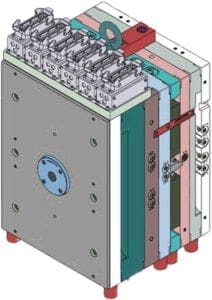

Maintenance of Your Tooling
Once your tooling is fully commercialised we deliver a maintenance program to ensure optimum results from your moulds. Any investment you make with Origin will normally include routine maintenance and replacement of some wearing parts and we will advise if any other parts should be held to ensure uninterrupted production of consistent quality components.
Project Confidentiality
Clients often choose to use Origin’s in-house services to ensure full protection of their intellectual property (IP) as we offer complete commercial confidence and security.
We work with a diverse range of global pharmaceutical and healthcare companies, many following conventional tooling development processes but also including others that have been fast-tracked to meet a specific need, or to remedy where a project has become derailed through some unforeseen event. Whatever your requirement, you can be sure of total confidentiality and protection of your IP. We can take a brief and extract much of the pressure from your team, working collaboratively to a greater or lesser degree – as you prefer.
- Collaboration
- Engineered with quality
- Component development
- Pilot mould development
- Optimised design and production
- Commercial production moulds
- Acceptance process
- Closed loop development and production
- Promoting a sustainable supply chain
Choose Origin for Expert Tooling Development and Design
With over 60 years of experience working with many of the world’s leading brand owners, we’re proud to offer our customers a range of custom-made solutions developed in our state-of-the-art cleanroom in the U.K.
Our facilities enable us to manufacture innovative thermoformed solutions, such as sterile barrier packaging, engineered to maximise revenue, reduce cost and increase your speed to market. No matter what you need when it comes to a cleanroom facility for tooling design and development, cleanroom manufacturing, cleanroom filling, or the packaging and labelling of medical and healthcare products, Origin Pharma Packaging will deliver a truly exceptional solution.